Subcortical Electrophysiology Mapping is a technique that pools data recorded from DBS electrodes across patients and maps this data onto anatomical space.
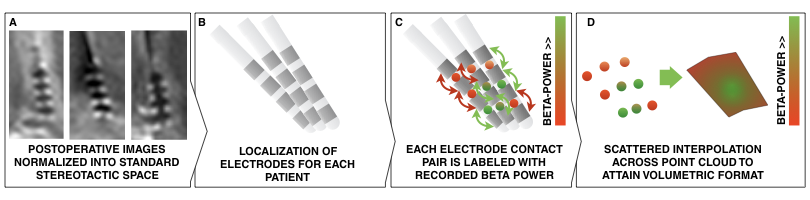
Methods summary. Postoperative images are transformed to standard space (A); electrodes are localized for each patient (B); electrophysiological recording estimates (such as beta power) are projected onto the midpoint of each contact pair (C); This leads to a 3D point cloud, each point associated with a beta power value. These points are used to fit a scattered interpolant to define beta values within covered subcortical space (D).
The methodology was successfully applied for the first time on beta-power recorded within the subthalamic nucleus in the two papers listed below.
Main findings of this paper were reproduced and extended by the following two studies of different labs:
Similarly, the method was applied to theta oscillations in the internal part of the pallidum in the following publication:
Finally, the concept was extended to electrophysiological features recorded during movement in this study:
The methodology is implemented in the Lead Group module supplied within Lead-DBS. The technique was summarized using the example of the same publication in our blog post here.
In theory, this mapping technique could be applied to virtually any measure that is prevalent for each patient, each DBS electrode, each DBS electrode contact or each DBS electrode contact pair. For instance, behavioral measures, electrophysiological recordings / task signatures, clinical outcome measures, patient demographical information, surgical techniques / treating DBS centers could be mapped to the subcortex using DBS electrode reconstructions from multiple patients.
For instance, a similar approach has been used to map clinical outcome (instead of electrophysiological measures) to the subcortex in these following (and other) studies. Some of the studies convolved DBS stimulation location by modeling a volume of activated tissue (VTA):